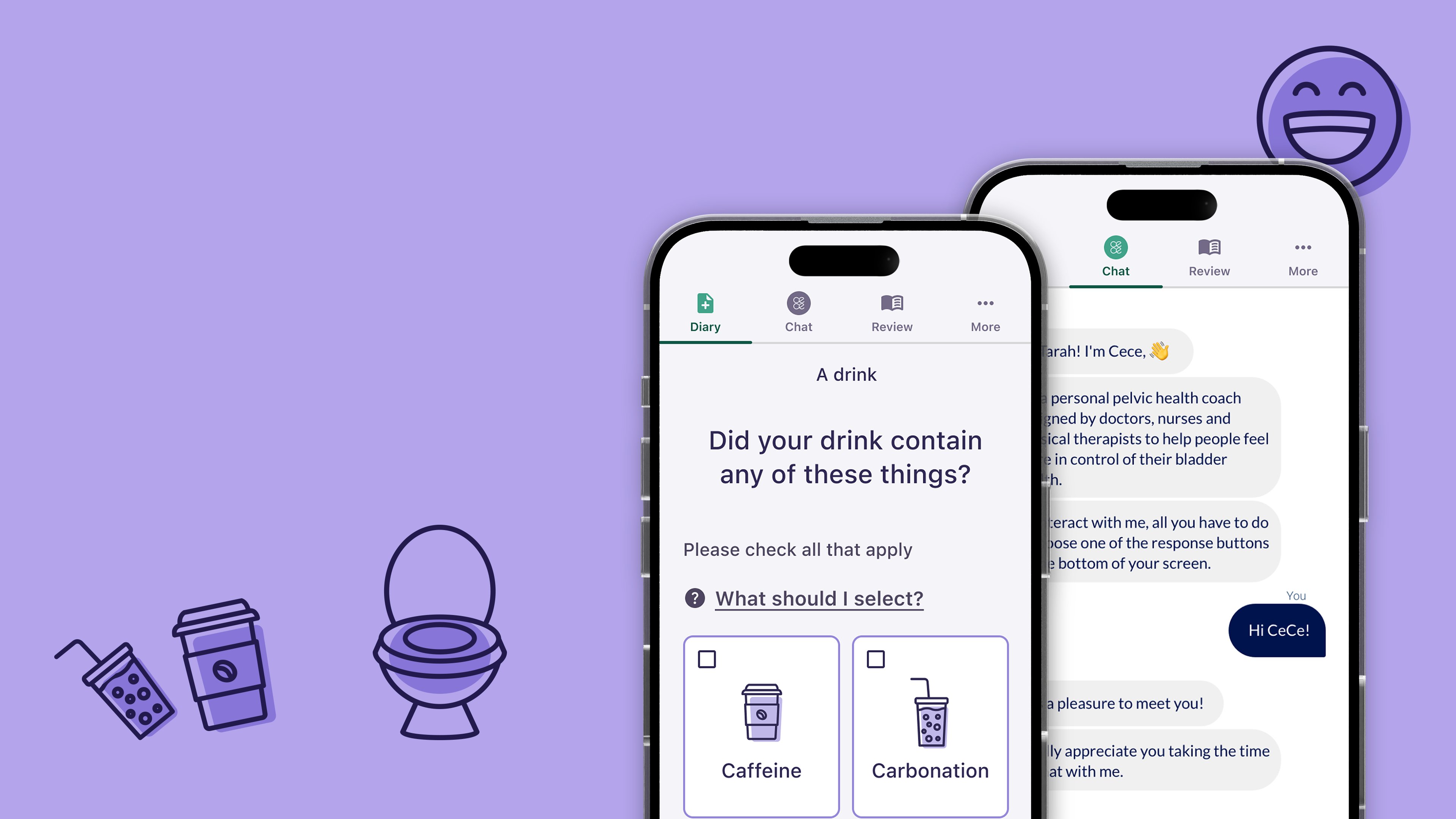
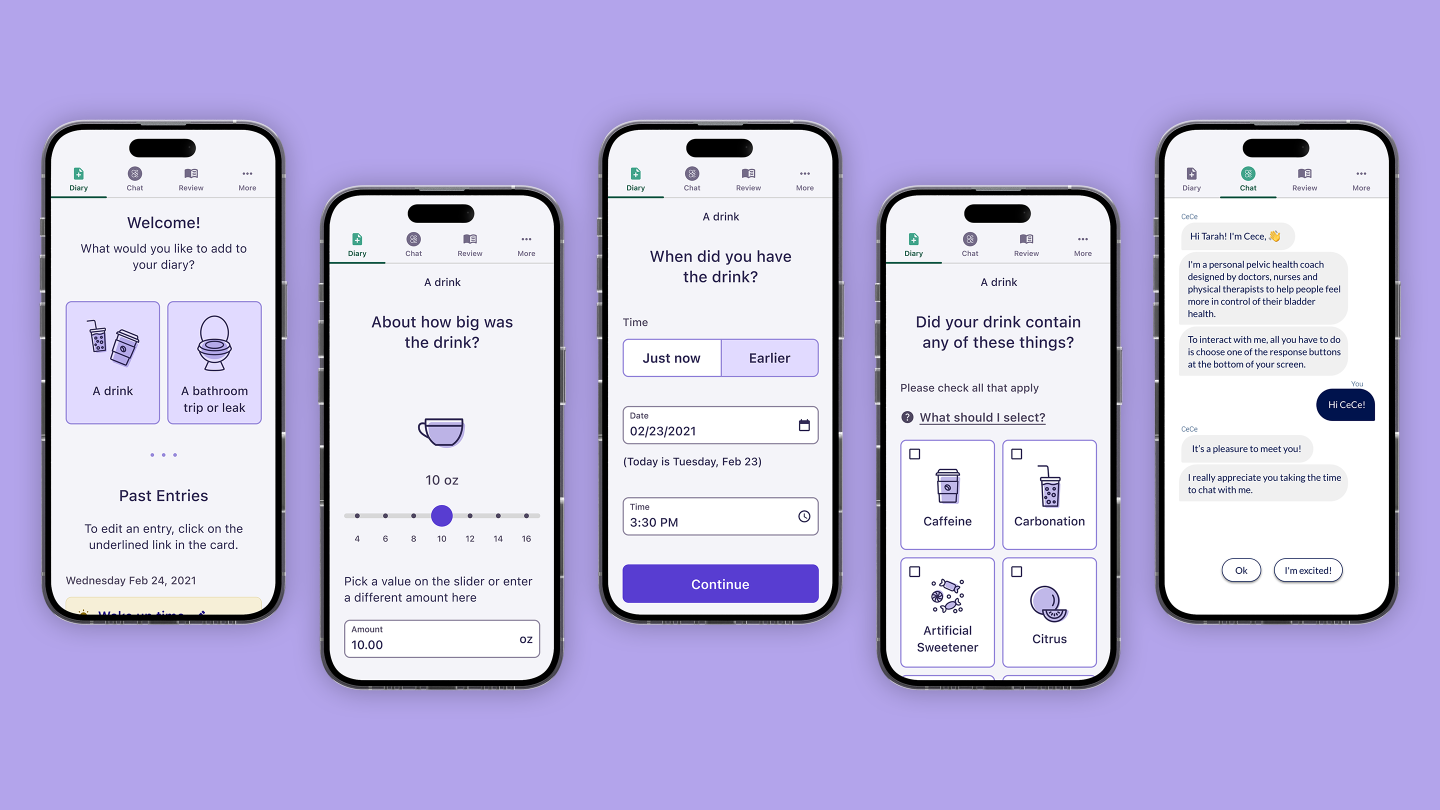
For people experiencing pelvic health disorders, the symptoms can be intensely embarrassing, and many are reluctant to identify and investigate their issues.
The opportunity to capture adequate patient health data and inform better avenues of treatment and education will come from an upcoming proof-of-concept study Renalis’ research partner, University Hospitals will lead.
The study will require participants to keep a bladder diary (tracking fluids in and fluids out) for three days. At the end of those three days, participants receive ongoing virtual behavioral therapy consisting of education and exercises to practice. There are two subsequent diaries at the four-week mark and eight-week mark, to show patients how and where they have improved over the course of the study.